Scientific Discoveries
There is an inverse regional relationship between the amount of tau and metabolism in the brain.
This finding does not mean that therapeutic approaches targeting amyloid are useless. They may well work in the pre-tau stage of the disease, when people predisposed to developing Alzheimer’s are still asymptomatic, but they are unlikely to help once tau has spread to neocortical networks. This assumption is supported by the recent announcements of the failure of amyloid-removing clinical trials at the early Alzheimer’s disease stages.
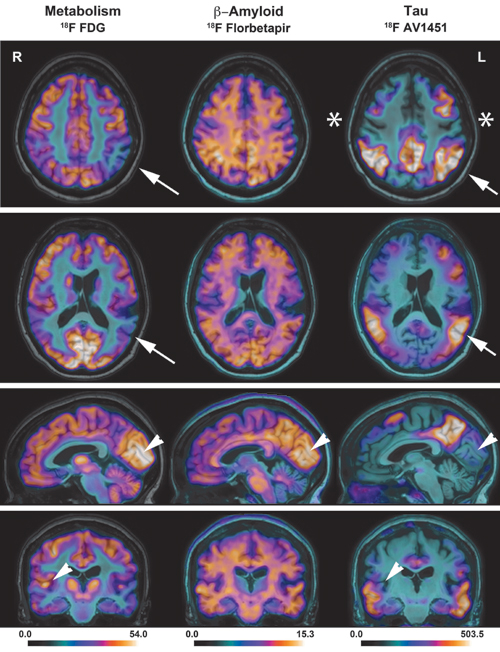
Fig 1. 18F-FDG, 18F-florbetapir, and 18F-AV-1451 PET superimposed to native MRI. Yin-yang relationship between tau and metabolism in the brain of a patient with Alzheimer’s disease. Multimodality positron emission tomography (PET) images are superimposed to native MRI. Primary sensory-motor areas (asterisks), as well as the primary visual (striatal cortex) and auditory (Heschl’s gyrus) regions (arrowheads) have normal metabolism and no tau deposition. Areas with high tau deposition (e.g. inferior parietal lobule, arrows) have decreased metabolism. This relationship does not apply to amyloid: notice high amyloid uptake in the calcarine cortex on the 18F-florbetapir scan (central column, arrowheads). AVID Radiopharmaceu-ticals provided the precursor for 18F-AV-1451.
Tau propagates within natural brain networks (“neurons that wire together degenerate together”).
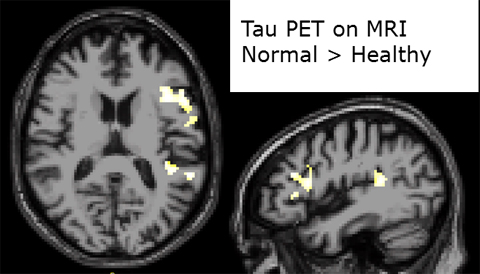
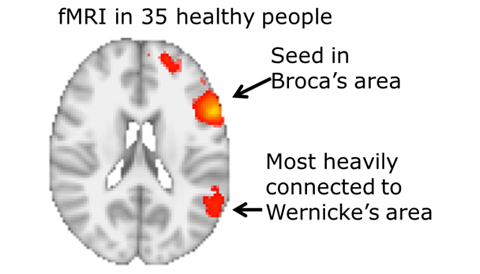
On voxel-wise analysis and compared to controls, patients with nfvPPA had increased tau uptake (p < 0.001) in two major clusters. The larger cluster was in Broca’s area. The smaller cluster was located in the planum temporale of the superior temporal gyrus, extending to the supramarginal gyrus (Wernicke’s area). As expected, MRI functional connectivity from the location of the larger tau cluster was greatest with a region located in the same area as the smaller tau cluster [4].
In older healthy subjects, [18F]AV-1451, which is supposed to bind to hyperphosphorylated tau, also seems to bind to regions (putamen and other nuclei) known not to have abnormal tau deposition on neuropathology (Fig 4). This so-called “off-target” binding has caused reports of abnormal tau deposition in tau PET studies of people who were actually normal. Our aim was to determine whether apparently increased specific [18F]AV-1451 binding in areas unlikely to harbor hyperphosphorylated tau, such as the substantia nigra, globus pallidus and putamen, of older subjects could be related to greater vascular permeability of these regions in older subjects as compared to younger ones.
For this purpose we measured dynamic [18F]AV-1451 uptake over a three-hour period in younger (23±2.1 years of age) and older (68.8±7.6 years) healthy subjects. In the same subjects we obtained dynamic gadolinium concentrations before and after a bolus injection of gadolinium, a large molecule commonly used to assess the permeability of the blood-brain barrier, by performing a pre-contrast 3D T1 map (five flip angle acquisitions) followed by a dynamic contrast enhanced MRI (DCE-MRI). During and after infusion, we acquired 180 consecutive T1-weighted volumes (3.4 sec per volume) over 10 minutes. Non-displaceable binding potential (BPND) of [18F]AV-1451 and permeability parameters (Ktrans) of gadolinium were analyzed by volume-of-interest methods and compared using Spearman’s correlation coefficients.
We found that there was greater BPND of [18F]AV-1451 in the globus pallidus and putamen (but not in cerebellum or cerebral cortex) of older as compared to younger subjects (Fig 4). Gadolinium concentration and permeability (Ktrans) was similar in the cerebellum, temporal cortex, and choroid plexus of younger and older subjects, but there was higher permeability in the substantia nigra, globus pallidus, and putamen (all p<0.05) of older as compared to younger subjects (Fig 5). Correlations between BPND and Ktrans were significant for substantia nigra, globus pallidus and putamen (all p<0.05) [5].
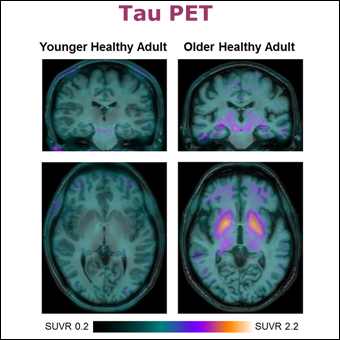
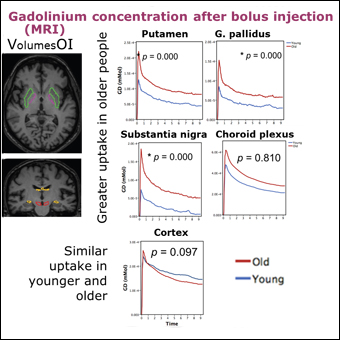
In summary, our work showed that increased vascular permeability in substantia nigra, pallidum and putamen of healthy older subjects could underlie some of the regional differences in [18F]AV-1451 uptake observed in older as compared to younger healthy subjects. These data are most relevant for the correct interpretation of the findings present in [18F]AV-1451 PET scans. This compound is now widely used to measure abnormal tau at all stages of the Alzheimer’s disease neurobiological continuum.
Most Recent Publications on Alzheimer’ Disease and Related Disorders (cited above):
- Masdeu, J. (2016) Misfolded tau, Alzheimer's bogeyman, exposed. JAMA Neurol in press.
- Pascual, B. and Masdeu, J.C. (2016) Tau, amyloid, and hypometabolism in the logopenic variant of primary progressive aphasia. Neurology 86, 487-488.
- Masdeu, J.C. and Pascual, B. (2016) Genetic and degenerative disorders primarily causing dementia. Handb Clin Neurol 135, 525-564.
- Pascual, B., Zanotti-Fregonara, P., Funk, Q., Rockers, E., Pal, N., Yu, M., . . . Masdeu, J. (2017) Nonfluent variant of primary progressive aphasia: Tau deposition in the language network. In Human Amyloid Imaging Conference.
- Pascual, B., Rockers, E., Bajaj, S., Yu, M., Karmonik, C., Xue, Z., and Masdeu, J. (2016) Older healthy people have increased vascular permeability in regions showing “off-target” [18F]AV-1451 uptake. In Alzheimer's Association International Conference.
