Scientists detect new ovarian cancer target, find existing drug to potentially treat it
Researchers at Houston Methodist Research Institute and The University of Texas MD Anderson Cancer Center have found a prescription drug, Calcitriol, approved by the Food and Drug Administration for the treatment of calcium deficiency and kidney diseases, may increase the likelihood of surviving ovarian cancer.
Their preclinical research, reported in the Journal of the National Cancer Institute, integrated computational modeling and biology experiments in cell lines and mouse models to pinpoint a molecular pathway between malignant cells and supportive cells, called fibroblasts, associated with poor prognosis for patients with high-grade serous ovarian cancer, the most common and difficult form of the disease.
A review of potential drugs by Houston Methodist researchers indicated that a synthetic and active form of a vitamin D analog called Calcitriol might break up molecular communication between cancer cells and fibroblasts, a finding confirmed in mouse models by MD Anderson researchers.
This study opens a new potential avenue for treating ovarian cancer. Since Calcitriol is an FDA-approved drug, no additional research is needed before the drug can advance to human clinical trials for ovarian cancer. The researchers are working to address regulatory procedures, planning and funding required to open a clinical trial.
“Targeting cancer cells might not be the only solution to treating cancer. Other cells in the tumor and surrounding microenvironment, such as fibroblasts, immune cells, fat cells and other supportive cells make up the very complex ecosystem of tumors that we need to understand,” said Samuel Mok, Ph.D., professor of Gynecologic Oncology and Reproductive Medicine and co-senior author of the study.
Using a computer modeling technique pioneered by co-senior author Stephen Wong, Ph.D., the Houston Methodist team processed and analyzed genomic big data to identify crosstalk targets and screen FDA-approved drugs. Their approach allowed them to look at the complexity of the interactions among tumor cells and different types of supporting cells to see a full map of their intricacies.
“In this era of big data, we can systematically identify pathways and therapies, as we’re using an unbiased approach to look at all possibilities,” said Wong, chair of systems medicine and bioengineering and John S. Dunn Sr. Presidential Distinguished Chair in Biomedical Engineering at Houston Methodist. “Our computational modeling can tell you which pathway is important for a particular disease. It allows for the heterogeneous tumor data to be fed in to the model to precisely identify these pathways, giving us a comprehensive view and allowing us to see specifically which malfunctioning cells to target.”
Fibroblasts make and maintain connective tissue that provides a scaffolding for organs. To better understand their role, Mok, co-lead author Tsz-Lun Yeung, Ph.D., and colleagues carved out fibroblasts from tumor samples to study separately, using a precise process called laser microdissection.
They studied gene expression by these cancer-associated fibroblasts (CAFs) and found the fibroblasts had two distinct expression, or signatures that they named CAF-C and CAF-N.
“A survival analysis of these two CAF types in 46 ovarian cancer patients showed that patients with CAF-C fibroblasts had a poor prognosis, with median overall survival of 16 months compared to 33 months for the other type,” Mok said.
Smad signaling between cancer cell, fibroblasts
The next step was to sort out what, specifically, makes the CAF-C group promote aggressive disease. Using a multi-cellular crosstalk modeling tool developed by Wong, called CCCExplorer (Cell-Cell Communication Explorer), co-lead author Jianting Sheng, Ph.D., and colleagues of the Houston Methodist team input gene-expression profiles from micro-dissected fibroblasts and neighboring ovarian cancer cells provided by Mok to compute, predict and prioritize crosstalk pathways and cell-to-cell interactions in the tumor microenvironment.
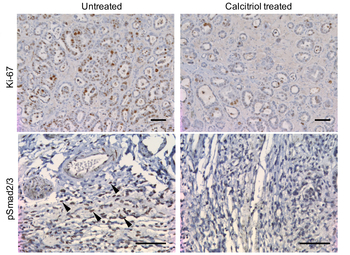
“We identified a signaling pathway, called Smad, as the culprit of poor ovarian cancer outcomes,” Wong said. “Reprogramming these cells by targeting their communication networks presents an opportunity for the development of new cancer treatment strategies. If we focus on targeting these supportive cells in the tumor microenvironment instead of the tumor, itself, it could lead to less toxic, more effective treatments.”
Experiments by Mok and colleagues confirmed the primacy of Smad signaling. Gene expression analysis again clustered patients into two groups; those with high expression of Smad-regulated genes had median overall survival of 15 months compared to 26 months for those with low expression.
Wong and colleagues at Houston Methodist applied another computational module in their CCCExplorer tool to predict known drugs that have passed phase I safety trials that might target Smad signaling. Calcitriol was selected for further studies in part because previous research shows it can inhibit the binding of Smad proteins to their target genes.
MD Anderson cell line experiments showed Calcitriol blocks Smad signaling. Treating ovarian-tumor bearing mice with Calcitriol reduced cancer cell proliferation and tumor volume while lengthening median overall survival from 36 to 48 weeks.
“We know that cells in the tumor microenvironment actually support the cancer and may contribute to its aggressiveness. This study opens up a new potential avenue for developing ovarian cancer treatments,” said co-author Karen Lu, M.D., chair of Gynecologic Oncology and Reproductive Medicine at MD Anderson and J. Taylor Wharton, M.D., Distinguished Chair in Gynecologic Oncology.
“These researchers have developed a unique and powerful concept to decode crosstalk and interactions among different cell components in the complex tumor microenvironment,” said Jenny C. Chang, M.D., director of Houston Methodist Cancer Center and Emily Hermann Chair in Cancer Research. “Instead of testing one hypothesis at a time, the modeling tool now allows the cancer researchers and drug designers to comprehensively evaluate major intercellular crosstalk pathways simultaneously to find novel targets for ovarian and other cancers.”
Co-authors with Mok, Wong, Yeung, Sheng and Lu are: Cecilia Leung, Ph.D., and Samuel Ho of Gynecologic Oncology and Reproductive Medicine at MD Anderson; Fuhai Li, Ph.D., of The Ohio State University College of Medicine, Columbus, Ohio; Jaeyon Kim, Ph.D., Melvin & Bren Simon Cancer Center, University of Indiana, Indianapolis; and Martin Matzuk, M.D., Ph.D., Baylor College of Medicine, Houston.
The study was funded by grants from the National Cancer Institute of the National Institutes of Health (R01CA133057, R01CA142832, RC4CA156551, U01188388, U54CA151668, U54CA149196, and UH2 TR000943) MD Anderson’s Ovarian Cancer Specialized Program of Research Excellence (SPORE) (P50CA083639), MD Anderson’s Uterine SPORE grant (P50CA098258) and by MD Anderson’s Cancer Center Support Grant (P30CA016672 ) from the National Institutes of Health; by the U.S. Department of Health and Human Services; the Ovarian Cancer Research Program, U.S. Department of Defense; the Gilder Foundation; the Cancer Prevention and Research Institute of Texas (CPRIT) and a CPRIT Core Facility Support Award: funding from Mr. Carl L. Norton, the Anna and John J. Sie Foundation, The Mary K. Chapman Foundation, the Ovarian Cancer Research Fund, the Ting Tsung and Wei Fong Chao Center for Bioinformatics Research and Imaging for Neurosciences (BRAIN), Cancer Fighters of Houston, Inc., and the John S. Dunn Foundation.
-------------------------------
Citation: Systematic Identification of Druggable Epithelial–Stromal Crosstalk Signaling Networks in Ovarian Cancer. Journal of the National Cancer Institute (June 1, 2018) T.-Lun Yeung, J. Sheng, C.S. Leung. F. Li, J.K. Samuel, Y. Ho, M.M. Matzuk, K.H. Lu, S.T.C. Wong, S.C. Mok.